We have studied the Delayed Luminescence (DL) both of mammalian cell cultures and skin in vivo in order to develop a new non-invasive diagnostic technique also for optical biopsy pourposes.
We have measured the DL emission of different cellular lines (normal, cancer and cancer-prone cell cultures) in presence or not of agents inducing apoptosis.The measurements performed on normal human fibroblast strains and human white melanoma cells have showed some of the most important DL physical characteristics.
-
DL emission is function of cellular lines and cellular densities. The differences between normal and tumor cells become evident at increasing of cellular density.
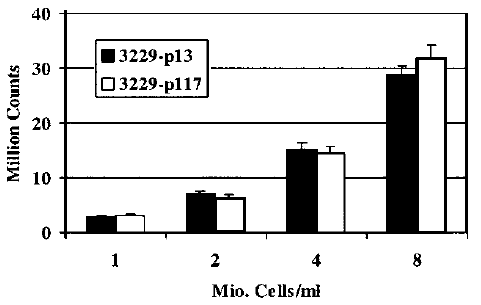 Figure 1 - Example of DL total numbers of emitted photons after UVA illumination, as a function of cell density. Normal young skin fibroblasts 3229: low passage number 13 (black), high p 117 (white) (Journal of Biomedical Optics 10(2), 024006 (2005))
Figure 1 - Example of DL total numbers of emitted photons after UVA illumination, as a function of cell density. Normal young skin fibroblasts 3229: low passage number 13 (black), high p 117 (white) (Journal of Biomedical Optics 10(2), 024006 (2005)) -
The temperature influence the DL emission spectra.
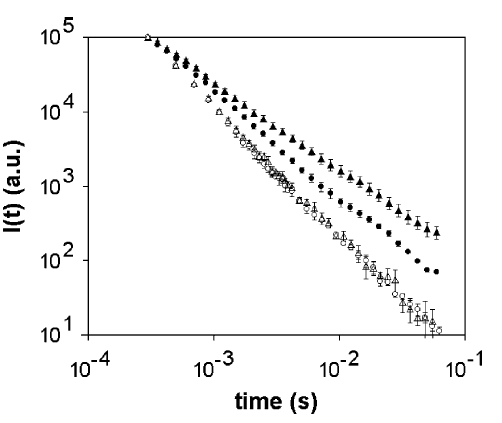
Figure 2 - Total DL dynamics of human cells samples at several temperatures after UV-light: ( ) human fibroblasts at 32 °C, ( ) human melanoma at 32 °C, ( ) human fibroblasts at 10 °C ( ) human melanoma a 10 °C. On raising the temperature the maximum of the spectral emission curve moves from the green (510 nm) to the yellow-orange (570 nm) region both for fibroblasts and melanoma cells. Moreover at low temperature the two spectra are, inside the experimental errors, nearly the same while at higher temperature they show measurable diversities, especially as it regards the relative intensities of the different components.
Two temperature have been checked: physiological temperature and a lower one. So we compared the ratio between every spectral component and the component at 460 nm for the two types of cells (normal vs. melanoma) at physiological temperature. Remarkably for the red component at 645 nm component the value of the ratio is 3.7 times greater in melanoma cells than in normal fibroblasts. In addition the ratio between the DL intensities at 567 nm and 509 nm shows an increase of about 30% from normal to tumor cells. Ulterior information can be gathered from the temporal trend of decays: as a matter of fact the different DL spectral components don't exhibit the same time trend. Starting from the experimental values of the intensity I(t) of luminescence we determined the values of the experimental decay probability P(t). It appeared that the P(t) function curves depend on the emission wavelength and/or the nature of the biological system. It was found that in the shorter time region (t < 100 ms), the curves relative to fibroblasts and melanoma cells are quite different and dependent on lemiss. As a matter of fact, the P(t) values of melanoma cells are about twice as that of fibroblasts for the spectral components with lemiss = 509 nm and l emiss > 550 nm. Moreover, in contrast to fibroblasts, in melanoma cells these components exhibit a marked maximum at about 30 ms. These results show evidence that the experimental technique used could be the basis for a new powerful non-invasive tool to determine biophysical changes within normal and tumor cells.
The results we obtained until now, have showeing a dependence of DL emission from the cellular line both in term of time trend and total count of the spectral components. Figures below show example of
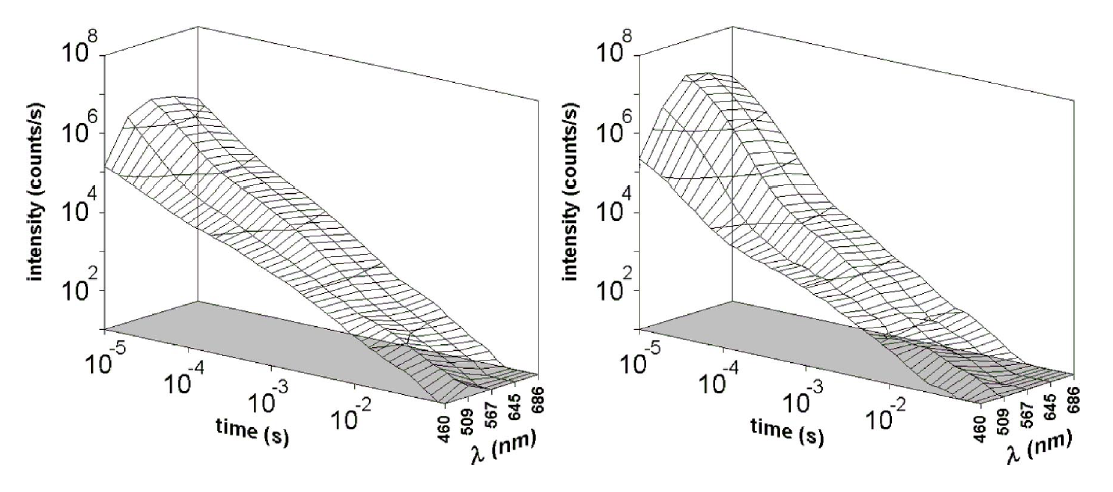
Figure 3 - Example of DL intensity as function of time and emission wavelenght: (on the left) human fibroblasts (on the right) human melanoma cells (Appl. Phys. Lett. 86, 153902 (2005))
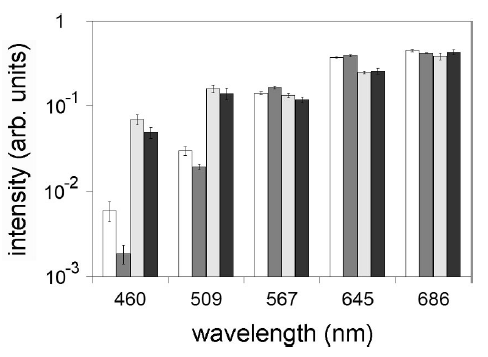
Figure 4 - Emission spectra of human fibroblast (HF) and human melanoma (HM) cells calculated averaging the actual spectra in two different time intervals. (white) HF, 10–50 µs; (dark grey) HM, 10–50 µs; (grey) HF, 10–50 ms (black); HM, 10–50 ms (Appl. Phys. Lett. 86, 153902 (2005))
Sorry... Under construction!